Immunohistochemistry is a specific protein stain that is used regularly throughout scientific research. Immunohistochemistry is a tissue stain that uses the specific binding of antibodies produced when an epitope (antigen or small segment of protein) is exposed to the immune cells of an animal from a different species. It involves the purification of a segment of a specific protein (epitope) that is to be investigated. This epitope is then injected to an animal that is a different species to the epitope host animal. After injection the animal will have an immune response to the epitope and will produce antibodies that will bind to the epitope specifically. These antibodies (referred to as the primary antibody) are then extracted from the animal’s serum and applied to the tissue that is to be stained for the epitope. The primary antibody solution is then washed off, leaving only the primary antibodies that are bound to the epitope on the tissue. The primary antibody will then be visualized by a number of methods.
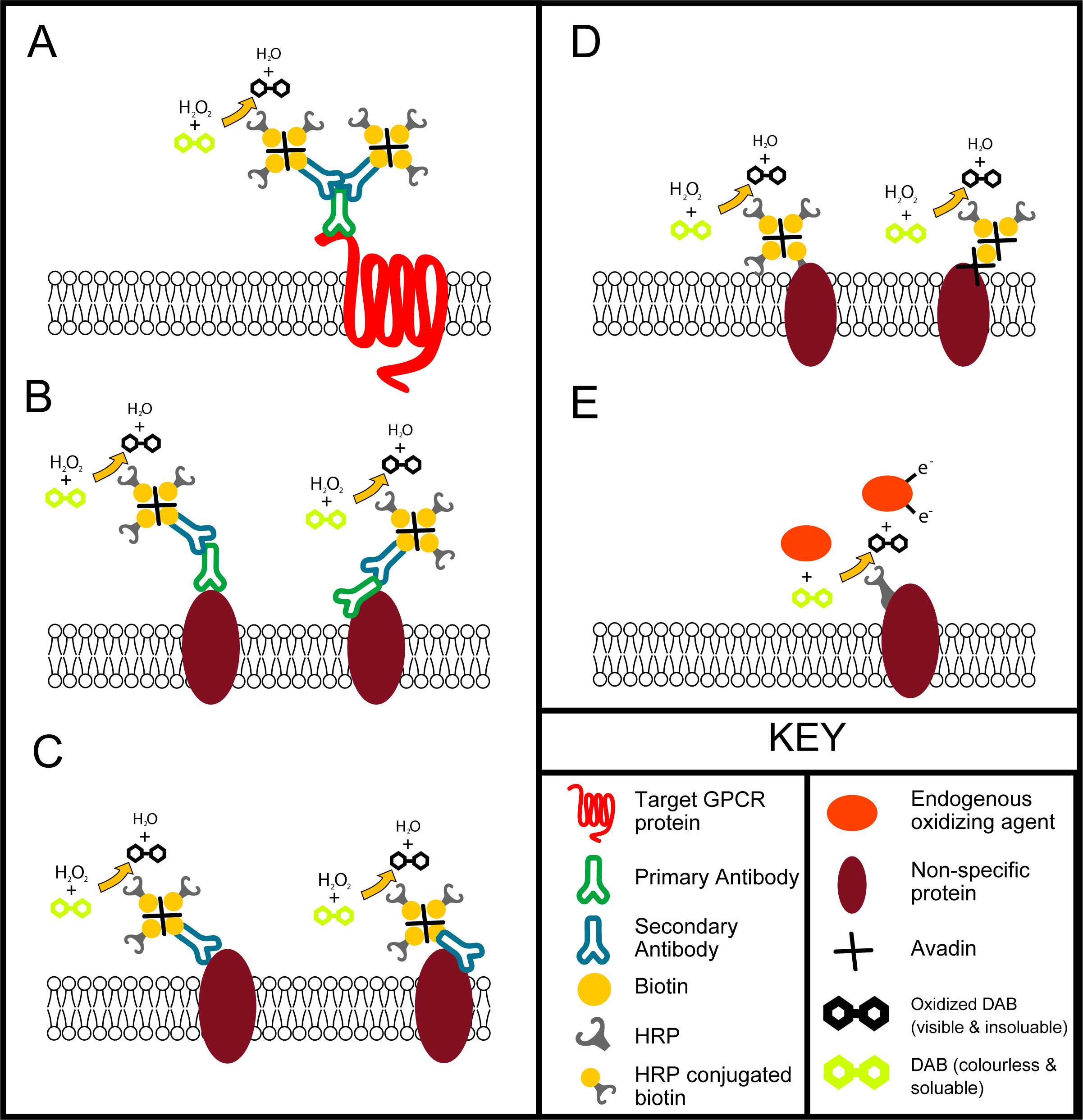
Problems occur when these antibodies bind to many proteins non-specifically. This can happen in a number of ways (see figure below), this leads to a miss-leading stain. There is huge debate in the literature about where the CBII receptor is, this is due largely to the unreliability of immunohistochemistry. We performed a study with 4 separate commercially bought CBII antibodies and one privately produced antibody. Using these we stained neurologically injured brains and found that one antibody showed large about of CBII expression on the injured side of the brain, another antibody showed only a small localized patch of staining right near the injury, yet another showed CBII expression over the entire brain, and the other two showed now staining at all. This demonstrated clearly the difficulties of immunohistochemistry and illustrated the need for the use of more than one technique (like in situ hybridization) and more than one antibody.
Ways in which labeling can occur in the tissue following immunohistochemistry. A- specific immunohistochemistry labeling of a target GRPC protein using primary antibody with a two biotinylated secondary antibodies attach, avidin has bound to the biotin on the secondary and three HRP conjugated biotins have bound to the avidins, the HRP has then facilitated the oxidation of colourless and soluble DAB into a darkened insoluble form. B- Nonspecific binding of the primary antibody to proteins on or within the cell, this can be binding of the binding domain (left) or binding of another region of the antibody (right). C- Nonspecific binding of the secondary antibody to proteins on or within the cell, this can be binding of the binding domain (left) or bind of another region of the antibody (right). D. Nonspecific binding of biotin (left) and avidin (right) to proteins within the tissue. E. Endogenous peroxidase activity that can oxidize DAB, this can be facilitated by membrane bound or free floating peroxidases. Illustrated by Jack Rivers